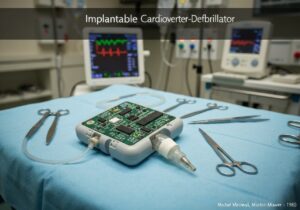
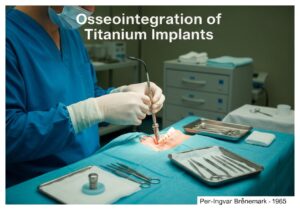
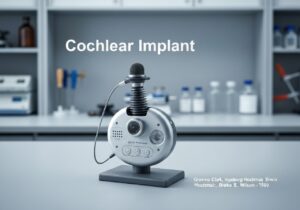
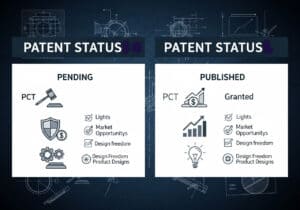
Medical devices are categorized into classes based on the risk they pose to patients and users. This risk-based approach, used by regulators like the US FDA and in the EU, typically involves three or four tiers (e.g., Class I, IIa, IIb, III). Low-risk devices like tongue depressors are Class I, while high-risk, life-sustaining devices like pacemakers are Class III.